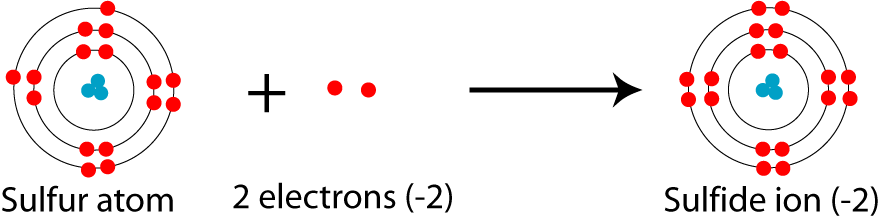
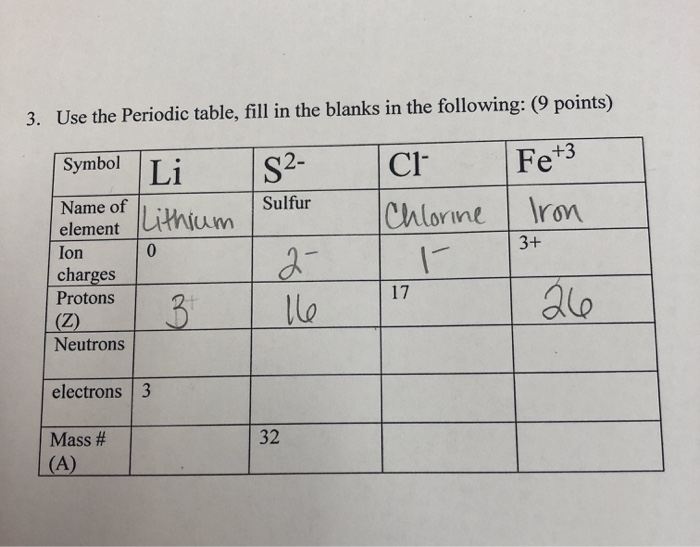
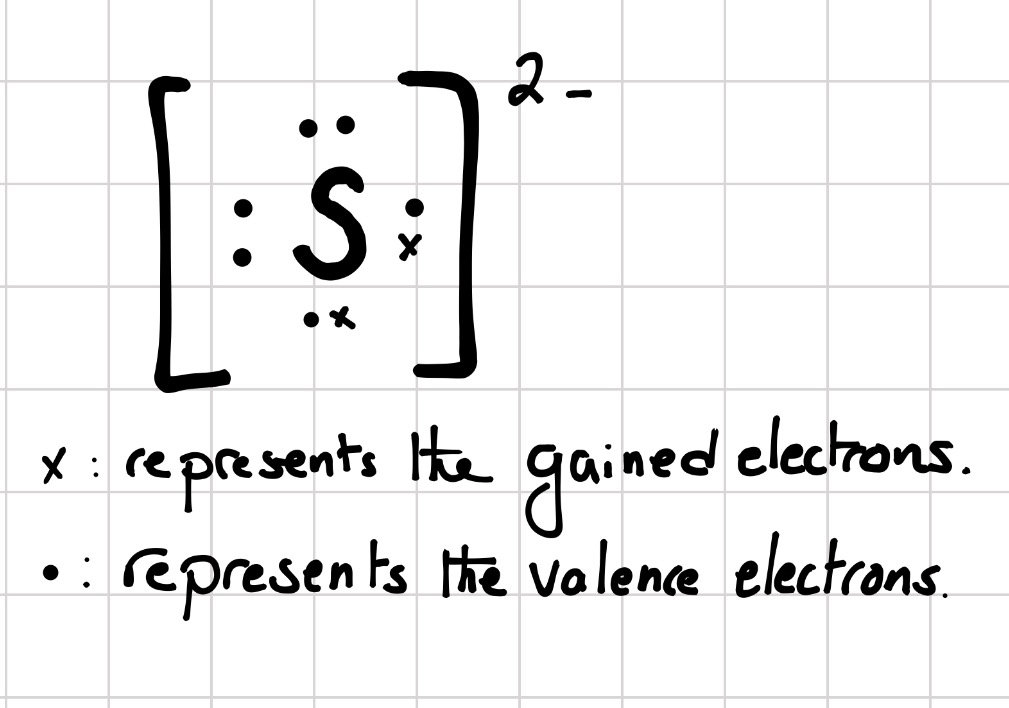
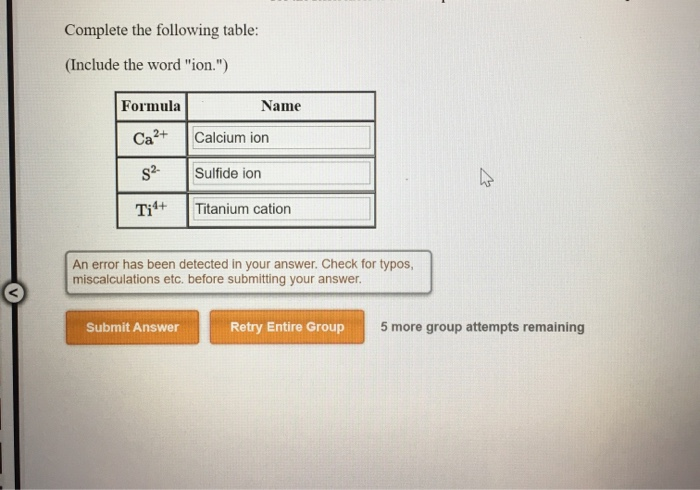
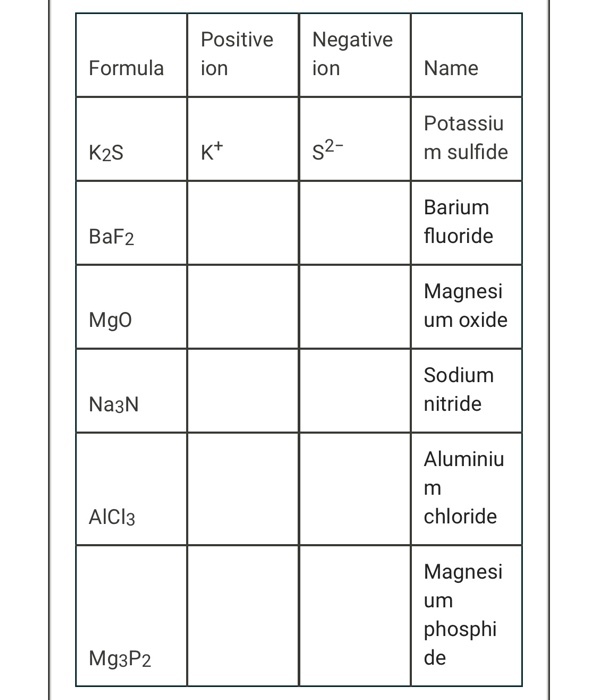
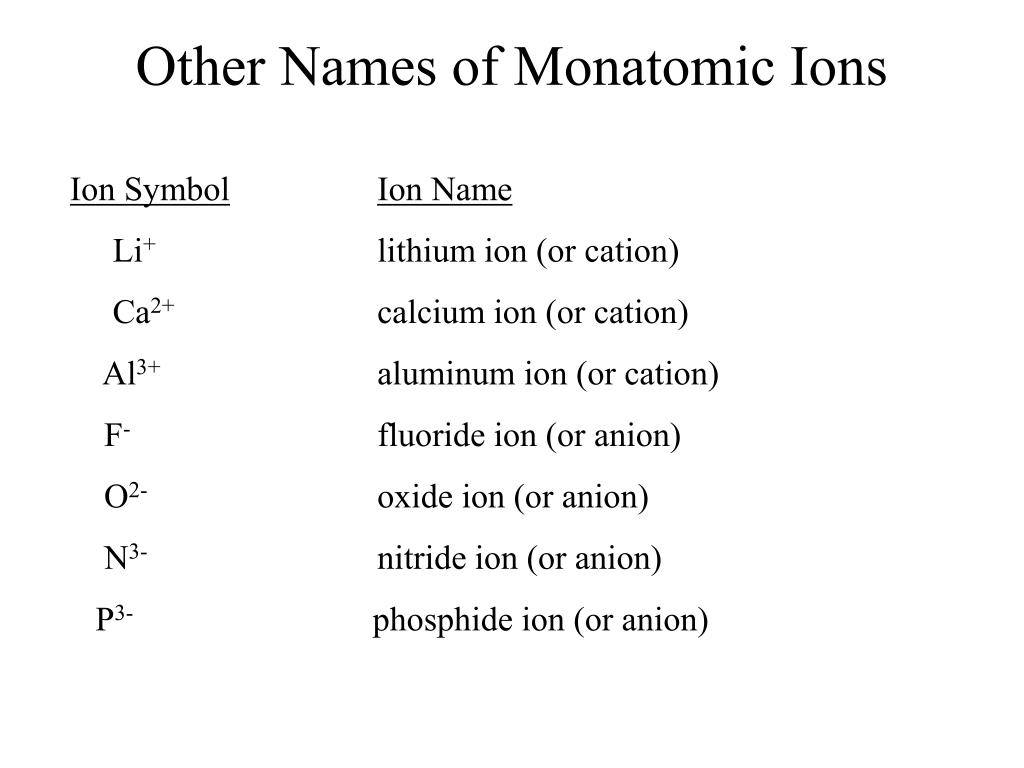
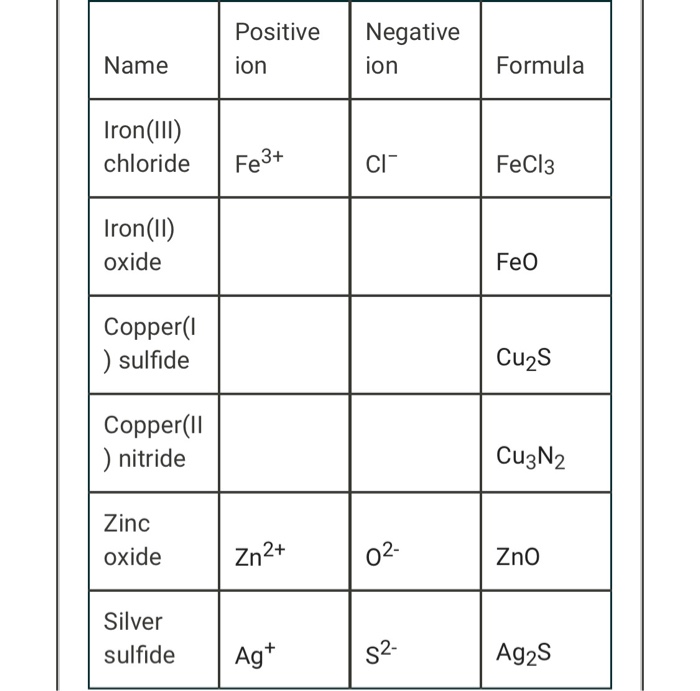
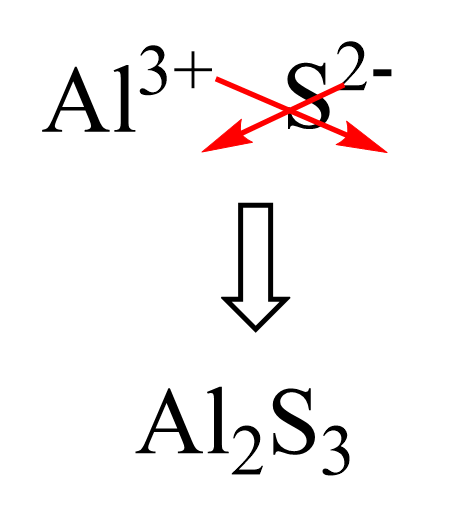
Chapter 5 Nomenclature Chemistry B2A. 1.Ionic compounds (a metal and a nonmetal) 2. Covalent compounds (two nonmetals) Binary Compounds. - ppt download
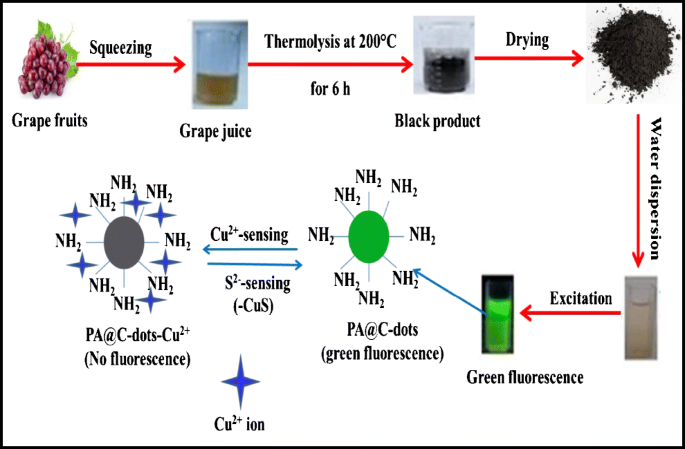
Development of dual function polyamine-functionalized carbon dots derived from one step green synthesis for quantitation of Cu2+ and S2− ions in complicated matrices with high selectivity | SpringerLink